SMT: Storage Of Solder Paste & Solderability
4. Storage of solder paste
Solder paste is shipped in jars ranging from 150 grams to 2 kilograms in size, in cartridges of 400 ml to 1.5 kilograms and in syringes of 10 to 100 grams. Storing the paste in a cold, dry area is beneficial. The cold temperature slows the rate of chemical reaction and reduces separation by significantly increasing the viscosity. The storage area should have a low controlled humidity (RH<50%) and the jars of solder paste should be tightly sealed. A refrigerator with a moisture absorbing desiccant works well for this purpose, unless specified otherwise by the paste supplier.
Reusing solder paste, from a previously opened jar or the excess from the printer, is acceptable if the paste is not separated and oxidized after exposure to moisture on the factory floor and subsequent storage. The reused paste should be tested for solder balls, slump and tacktime before reusing.
5. Solderability
Solderability is the property of wetting. Good wetting results in the formation of a uniform, smooth, unbroken, adherent coat of solder on the base metal. Therefore, wetting is an essential prerequisite for soldering. Wetting means that molten solder diffuses into the solid surface of the metal to be soldered. Wetting is possible only if the solder can come into immediate contact with the metallic surface of the solid metal part. Any filmy adhering contamination such as oxides, oils on the surface to be soldered will act as barrier to metallic continuity and thus prevent wetting. A drop of solder on a contaminated surface behaves in the same way as an isolated drop of water on a greasy plate. See fig. below.
Left: Drop of oxidized plate; the solder doesn’t wet. Right: Drop on a clean plate; the solder spreads while a diffusion layer grows at the interface.
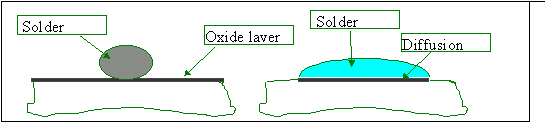
The kind of joint in which an oxide layer is still present between the base metal and the solder is obtained if one works at too low a temperature. Such joints are called cold joints. With poor solderability, non-wetting and de-wetting are observed. In nonwetting, the solder does not adhere to the surface to be soldered. This condition, the opposite of wetting, is caused by a physical barrier (intermetallic or oxide) between the solder and the base metal. The predominant cause is the total consumption of the layer of solder, which turns into intermetallic compound of the base metal. Good solderability can be achieved by pretinning all incoming parts that don’t meet the solderability requirements using an active flux. Dewetting is a condition that occurs when molten solder has coated a surface and then receded, leaving irregularly shaped mounds of solder separated by areas covered with a thin solder film.
Solderability of components wired boards can be tested visually by edge-dip method.
